Determination of Specification of Medical Use Invention
I. Appeal against Decision to Reject Patent Application
No. 2000-7002144
Patent Court’s Decision No. 2004HEO4525 published on Apr. 29, 2005
Supreme Court’s Decision No. 2005HU1417 published on Mar. 30, 2007
- dismissed
1. Present patent invention (claim 1)
A pharmaceutical composition to control angina pectoris, atherosclerosis, combined hypertension and hyperlipidemia and symptoms of cardiac risk, comprising:
(a) an amount of amlodipine or a pharmaceutically acceptable acid addition salt thereof ;
(b) an amount of atorvastatin or a pharmaceutically acceptable salt thereof; and
(c) a pharmaceutically acceptable carrier or diluent.
2. Patent Court’s decision (2004HEO4525)
Even though the individual pharmacological mechanisms of amlodipine and atorvastatin which are activating elements of the composition according to the present patent invention, when amlodipine is used with atorvastatin, it is not known which chemical reaction is made by these elements and which interaction is made by these elements. Therefore, when amlodipine is used with atorvastatin, it cannot be concluded that the pharmacological mechanism corresponding to the sum of the respective pharmacological mechanisms of amlodipine and atorvastatin is naturally generated. As mentioned above, what is publicly known before the priority date of the present invention as filed is not the pharmacological mechanisms according to the use of a calcium channel blocker with a HMG-CoA reductase inhibitor as the superordinate concept but the pharmacological mechanisms of amlodipine and atorvastatin which are ones of the calcium channel blocker and the HMG-CoA reductase inhibitor. Many compounds are used as the calcium channel blocker and the HMG-CoA reductase inhibitor. When so many compounds are used together, it cannot be concluded that the same pharmacological mechanisms are generated. Thus, it is difficult to consider that the pharmacological mechanisms according to the use of amlodipine with atorvastatin indicating the pharmaceutical efficacy of the compounds described in the claims of the present invention are clearly proved before the priority date of the present invention..
When the effects of the invention are not explicitly described in the specification but the effects of the invention can be inferred from the description of the purpose and constitution of the invention, an argument regarding the effects is allowed to be submitted later. Where the pharmacological mechanisms of an invention are explicitly described in the specification like the present patent invention but, since the pharmacological mechanisms indicating the pharmaceutical efficacy described in the specification is not clearly proved, the point that the compositions have the pharmaceutical efficacy as described is required to be described by providing the examples with pharmacological data or the like or a detailed description capable of replacing the examples, the later submission of the quantitative pharmacological data regarding the pharmaceutical efficacy is not allowed.
Therefore, it cannot be considered that the pharmacological mechanisms indicating the pharmaceutical efficacy described in the specification is clearly proved before the priority date of the present patent invention. Further, the detailed description of the invention does not provide any concrete description which is capable of replacing the quantitative pharmacological data of the pharmaceutical efficacy. Therefore, the present patent invention is not considered as complying with the requirements that the specification shall describe the effects or the like of the invention so that any person skilled in the art can easily carry out the invention.
3. Supreme Court’s decision (2005HU1417)
(1) Specification descriptive requirements of for a medical use invention
In the specification attached to a patent application, a ‘detailed explanation’ of an invention shall describe the invention in terms of the purpose, constitution and effects, so that a person having ordinary knowledge in the art field to which the invention pertains can correctly understand the invention and reproduce it from the description in the specification based on the technical level at the time of filing the application, without any addition of special knowledge. Specifically, in a medical use invention where the description of pharmaceutical efficacy is required, unless there is any special circumstances, like that the pharmacological mechanisms indicating the pharmaceutical efficacy described in the specification are clearly proved before the filing of the application, it is required to describe that a particular substance has the pharmaceutical efficacy by providing an example with pharmacological data or the like or a concrete description capable of replacing the example. Only when these requirements are satisfied, the invention is considered as being completed and complying with the descriptive requirements in the specification.
(2) Regarding the present patent invention described in claim 1, the respective pharmacological mechanisms of amlodipine and atorvastatin which are individual activating elements are separately publicly known before the priority date of the present invention, and the fact that the use of amlodipine and atorvastatin is effective for the disease such as angina pectoris is publicly known. However, by the reasons of these facts, the pharmacological mechanisms of the composition of amlodipine and atorvastatin according to the present invention described in claim 1 cannot be considered as being clearly proved. Nevertheless, the specification provides neither any examples including pharmacological data or the like regarding the pharmaceutical efficacy of the composition nor any concrete descriptions capable of replacing the examples. Therefore, the pharmaceutical efficacy of the composition according to the present invention cannot be considered as being clearly described so that the person having ordinary knowledge in the art field to which the invention pertains can easily understand the invention. Further, regarding the present invention described in claim1, the pharmacological mechanisms of which are not clearly proved, it is not allowed to submit the pharmacological data or the like to prove the pharmaceutical efficacy after the filing of the application.
II. Appeal against Decision to Reject Patent Application No. 1995-703700
Patent Court’s Decision No. 2002HEO4682 published on Jun. 5, 2003
Supreme Court’s Decision No. 2003HU1550 published on Dec. 23, 2004
- dismissed
1. Gist of the present patent invention (claim 1)
A composition to effectively inhibit angiogenesis, comprising:
an angiogenesis-inhibiting compound having the following formula; and
a pharmaceutically acceptable carrier.
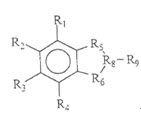
(Since the substituent is not related to the point at issue, the description of the substituent is omitted.)
2. Patent Court’s decision (2002HEO4682)
(1) Specification descriptive requirements of medical use invention
According to Article 42(3) of the Patent Law, a ‘detailed explanation of an invention’ in the specification to be submitted with a patent application shall describe the invention in terms of the purpose, constitution and effects of the invention so that a person having ordinary knowledge in the art field to which the invention pertains can easily carry out the invention. The phrase ‘so that a person having ordinary knowledge in the art field to which the invention pertains can easily carry out the invention’ means that a person having a technical understanding at a common level in the art field to which the invention pertains, i.e., an average skilled person, can correctly understand the invention and reproduce the invention from the descript
|